At UNSW Canberra
Synthesis and spectroscopic investigation of substituted piperazine-2,5-dione derivatives
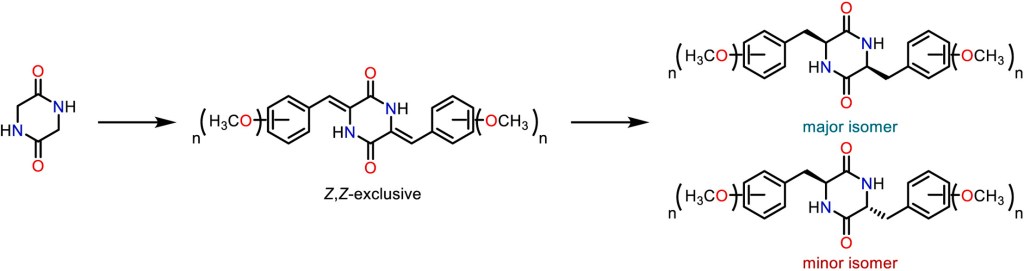
Tetrahedron, 2024, 153, 133838. 10.1016/j.tet.2024.133838
The pyrazole derivative of usnic acid inhibits the proliferation of pancreatic cancer cells in vitro and in vivo

Cancer Cell International, 2023, 23, 210. 10.1186/s12935-023-03054-x
Synthetic cannabinoid receptor agonists: An overview
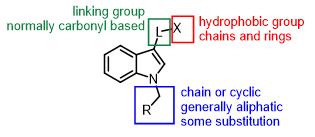
Neurobiology and Physiology of the Endocannabinoid System, 2023, 493. 10.1016/B978-0-323-90877-1.00038-3
Divergent reactivity of usnic acid and evaluation of its derivatives for antiproliferative activity against cancer cells

Bioorganic & Medicinal Chemistry, 2023, 79, 117157. 10.1016/j.bmc.2023.117157
The Isoxazole Derivative of Usnic Acid Induces an ER Stress Response in Breast Cancer Cells That Leads to Paraptosis-like Cell Death
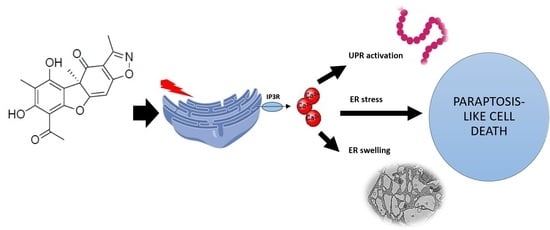
International Journal of Molecular Sciences, 2022, 23(3), 1802. 10.3390/ijms23031802
Prior to UNSW Canberra
The Current Status of Drug Discovery for the Oxytocin Receptor
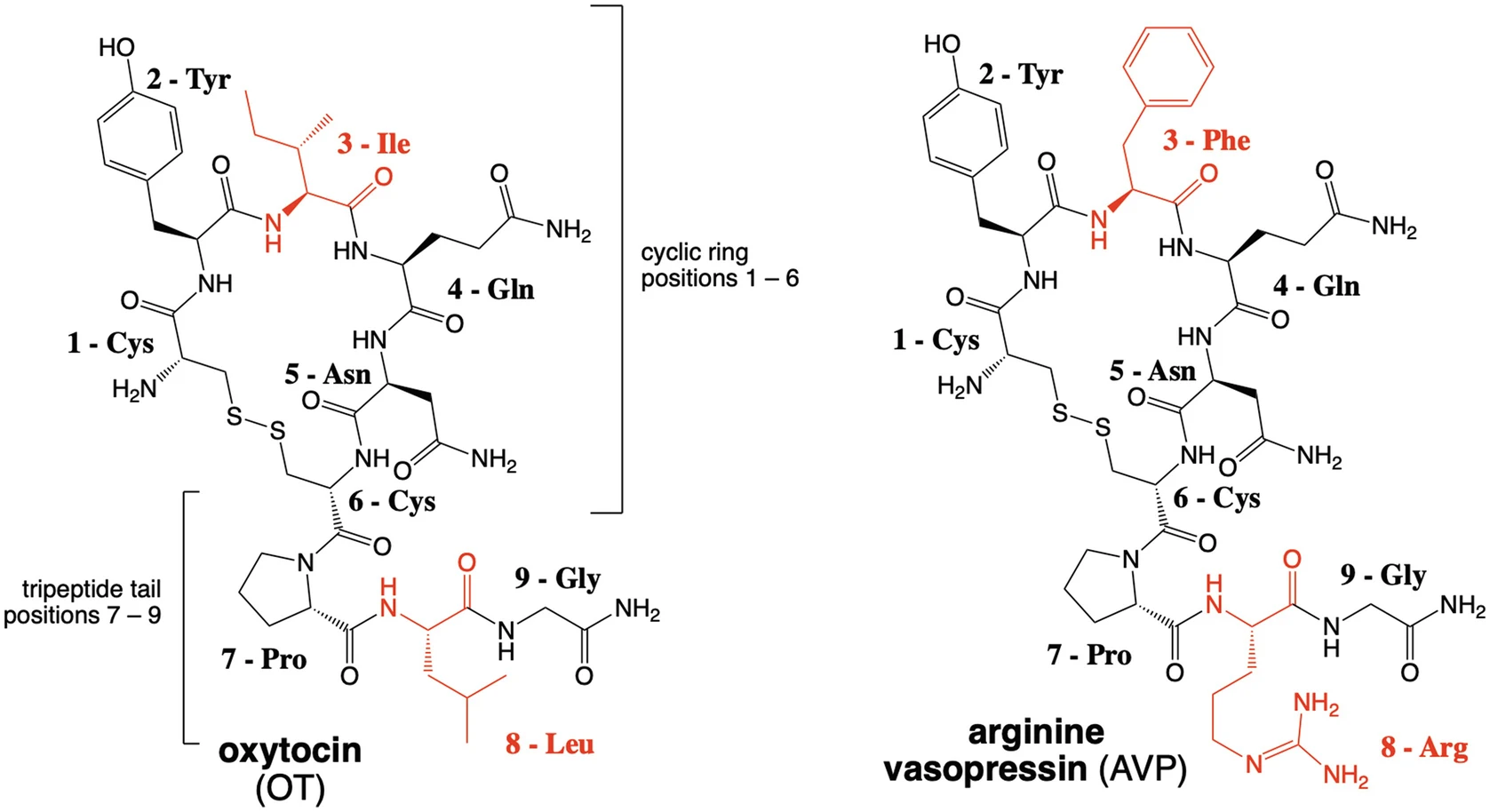
Book chapter in Oxytocin: Methods in Molecular Biology. 10.1007/978-1-0716-1759-5_10
Tobramycin and Colistin display anti-inflammatory properties in CuFi-1 cystic fibrosis cell line

European Journal of Pharmacology, 2021, 902, 174098. 10.1016/j.ejphar.2021.174098
Prodromal neuroinflammatory, cholinergic and metabolite dysfunction detected by PET and MRS in the TgF344-AD transgenic rat model of AD: a collaborative multi-modal study
Theranostics, 2021, 11(14), 6644. 10.7150/thno.56059
Tricyclic heterocycles display diverse sensitivity to the A147T TSPO polymorphism

European Journal of Medicinal Chemistry, 2020, 207, 112725. 10.1016/j.ejmech.2020.112725
Rapid Antibacterial Activity of Cannabichromenic Acid against Methicillin-Resistant Staphylococcus aureus
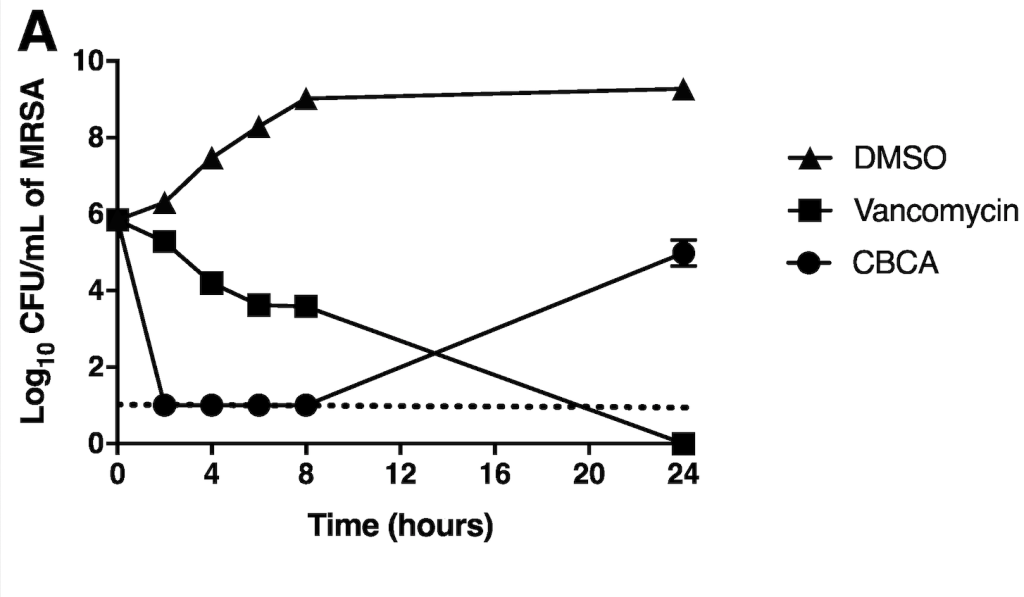
Antibiotics, 2020, 9(8), 523. 10.3390/antibiotics9080523
Targeting the MAPK7/MMP9 axis for metastasis in primary bone cancer
Oncogene, 2020, 39, 5553. 10.1038/s41388-020-1379-0
Low intrinsic efficacy for G protein activation can explain the improved side effect profiles of new opioid agonists

Science Signaling, 2020, 13, eaaz3140. 10.1126/scisignal.aaz3140
Reversing binding sensitivity to A147T translocator protein
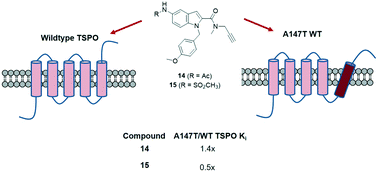
RSC Medicinal Chemistry, 2020, 11, 511. 10.1021/10.1039/C9MD00580C
First Nondiscriminating Translocator Protein Ligands Produced from a Carbazole Scaffold
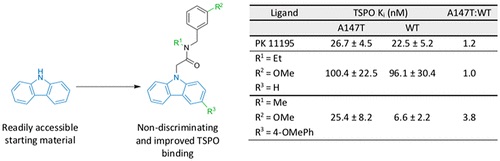
Journal of Medicinal Chemistry, 2019, 62, 8235. 10.1021/acs.jmedchem.9b00980
An overview of late-stage functionalization in today’s drug discovery
Expert Opinion on Drug Discovery, 2019, 14, 1137. 10.1080/17460441.2019.1653850
Synthesis of Usnic Acid Derivatives and Evaluation of Their Antiproliferative Activity against Cancer Cells

Journal of Natural Products, 2019, 82, 1768. 10.1021/acs.jnatprod.8b00980
Challenges and Opportunities in Central Nervous System Drug Discovery
Trends in Chemistry, 2019, 1, 612. 10.1016/j.trechm.2019.04.009
New-generation azaindole-adamantyl-derived synthetic cannabinoids
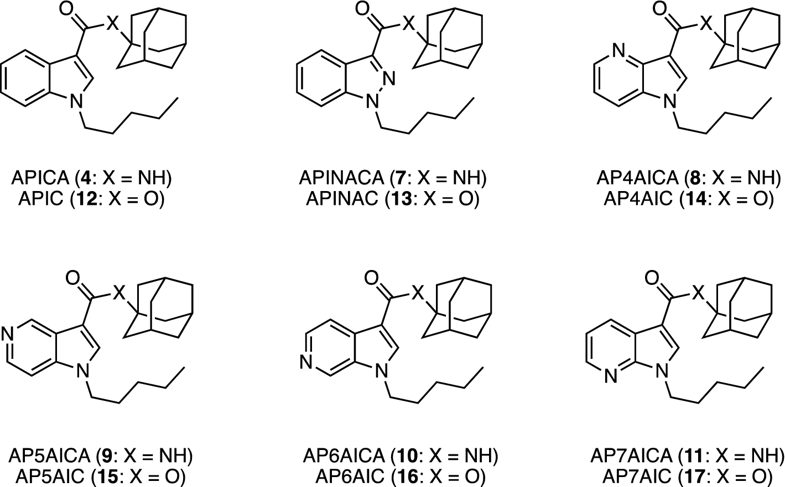
Forensic Toxicology, 2019, 37, 350. 10.1007/s11419-019-00466-1
Imaging glial activation in patients with post-treatment Lyme disease symptoms: a pilot study using [11C]DPA-713 PET

Journal of Neuroinflammation, 2018, 15, 346. 10.1186/s12974-018-1381-4
In vivo assessment of neuroinflammation in progressive multiple sclerosis: a proof of concept study with [18F]DPA714 PET
Journal of Neuroinflammation, 2018, 15, 314. 10.1186/s12974-018-1352-9
Synthesis and in vitro evaluation of diverse heterocyclic diphenolic compounds as inhibitors of DYRK1A

Bioorganic & Medicinal Chemistry, 2018, 26, 5852. 10.1016/j.bmc.2018.10.034
Targeting the Oxytocin System: New Pharmacotherapeutic Approaches

Trends in Pharmacological Sciences, 2018, 40, 22. 10.1016/j.tips.2018.11.001
Flexible Analogues of Azaindole DYRK1A Inhibitors Elicit Cytotoxicity in Glioblastoma Cells

Australian Journal of Chemistry, 2018, 71(10), 789. 10.1071/CH18251
Cubanes in Medicinal Chemistry

Journal of Medicinal Chemistry, 2019, 62, 1078. 10.1021/acs.jmedchem.8b00888
Peptides, Peptidomimetics, and Carbohydrate–Peptide Conjugates as Amyloidogenic Aggregation Inhibitors for Alzheimer’s Disease

ACS Chemical Neuroscience, 2018, 9, 1530. 10.1021/acschemneuro.8b00185
The role of polycyclic frameworks in modulating P2X7 receptor function

Tetrahedron, 2018, 74, 1207. 10.1016/j.tet.2017.10.075
Conformationally rigid derivatives of WAY-267,464: Synthesis and pharmacology at the human oxytocin and vasopressin-1a receptors
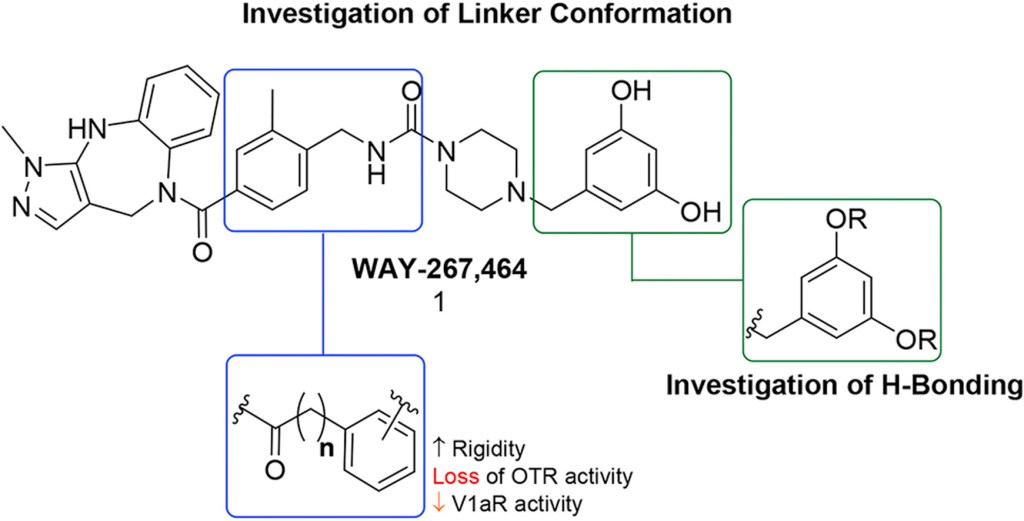
European Journal of Organic Chemistry, 2018, 143, 1644. 10.1016/j.ejmech.2017.10.059
The evolving science of phytocannabinoids

Nature Reviews Chemistry, 2017, 2, 0101. 10.1038/s41570-017-0101
Investigation of pyrazolo-sulfonamides as putative small molecule oxytocin receptor agonists
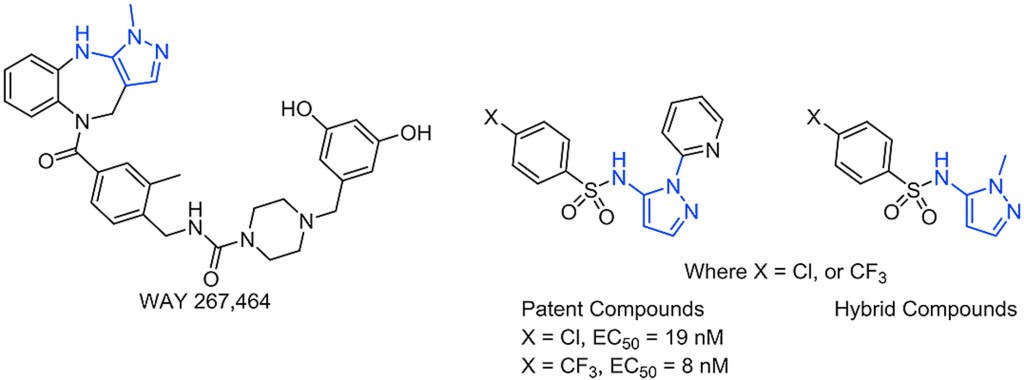
European Journal of Medicinal Chemistry, 2017, 136, 330. 10.1016/j.ejmech.2017.05.013
Metal-acetylide addition to tetracyanoethylene

Tetrahedron Letters, 2017, 58, 2414. 10.1016/j.tetlet.2017.05.009
Pharmacological evaluation of a novel series of urea, thiourea and guanidine derivatives as P2X7receptor antagonists

Bioorganic & Medicinal Chemistry Letters, 2017, 27, 2439. 10.1016/j.bmcl.2017.04.005
Porphyrin Donor and Tunable Push–Pull Acceptor Conjugates—Experimental Investigation of Marcus Theory

Chemistry–A European Journal, 2017, 23, 6357. 10.1002/chem.201700043
Discovery and pharmacological evaluation of a novel series of adamantyl cyanoguanidines as P2X7 receptor antagonists
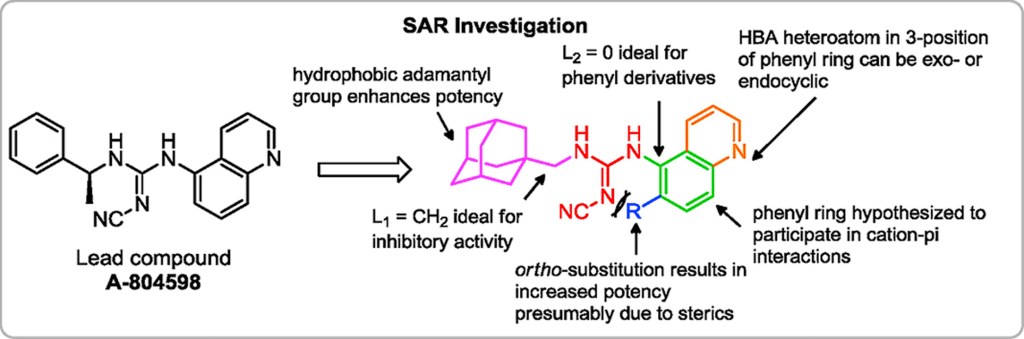
European Journal of Medicinal Chemistry, 2017, 130, 433. 10.1016/j.ejmech.2017.02.060
Structural Optimization and Pharmacological Evaluation of Inhibitors Targeting Dual-Specificity Tyrosine Phosphorylation-Regulated Kinases (DYRK) and CDC-like kinases (CLK) in Glioblastoma

Journal of Medicinal Chemistry, 2017, 60, 2052. 10.1021/acs.jmedchem.6b01840
Comparative Evaluation of Three TSPO PET Radiotracers in a LPS-Induced Model of Mild Neuroinflammation in Rats
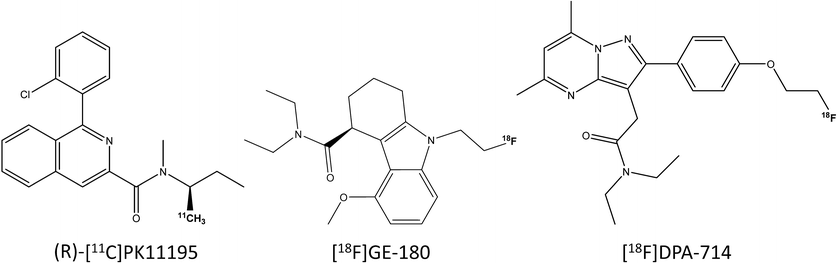
Molecular Imaging and Biology, 2017, 19, 77.
Ring-opened aminothienopyridazines as novel tau aggregation inhibitors

Medicinal Chemistry Communications, 2017, 8, 1275. 10.1039/C6MD00306K
Rapid access to N-(indol-2-yl)amides and N-(indol-3-yl)amides as unexplored pharmacophores
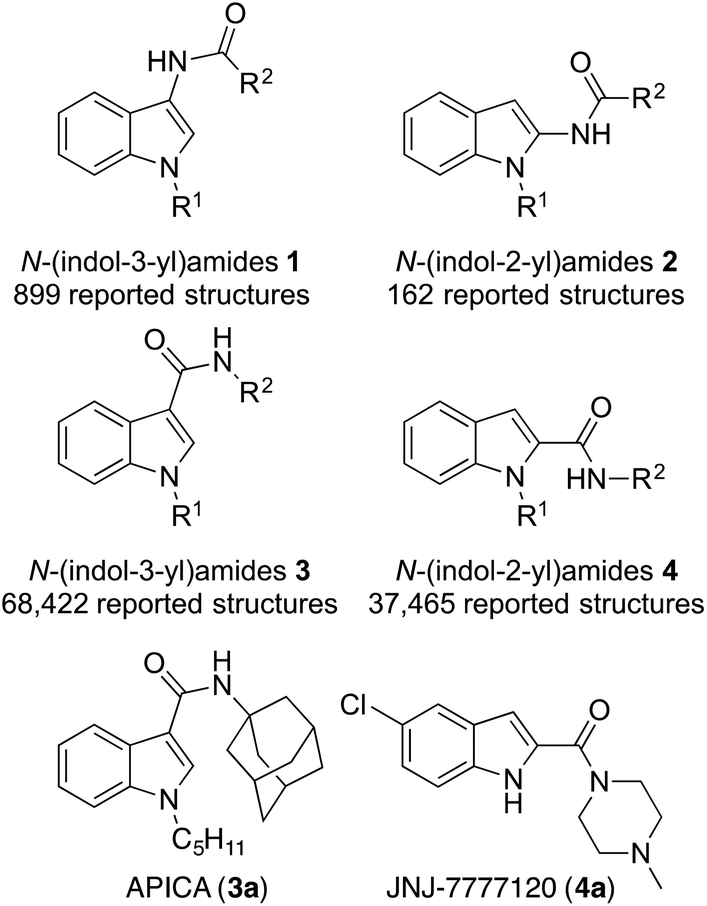
Organic & Biomolecular Chemistry, 2017, 15, 576. 10.1039/C6OB02622B
Determination and reduction of translocator protein (TSPO) ligand rs6971 discrimination
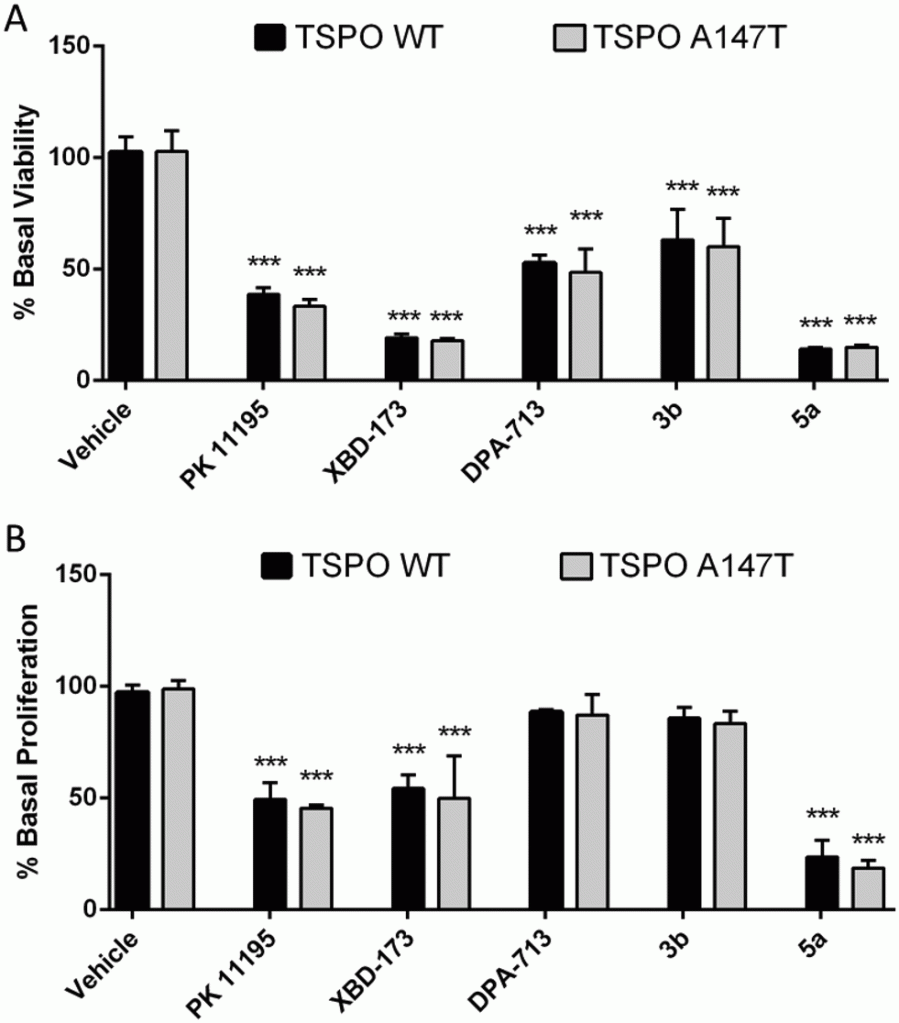
Medicinal Chemistry Communications, 2017, 8, 202. 10.1039/C6MD00523C
The Formation of Seven-Membered Heterocycles under Mild Pictet–Spengler Conditions: A Route to Pyrazolo[3,4]benzodiazepines
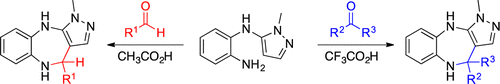
Journal of Organic Chemistry, 2016, 81, 4883. 10.1021/acs.joc.6b00710
A Three-Step Synthesis of Tetrasubstituted NH-Pyrroles
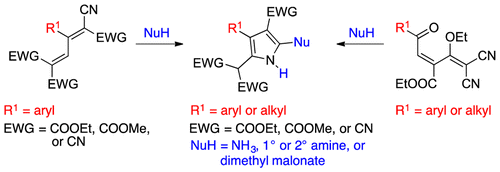
Organic Letters, 2016, 18, 2252. 10.1021/acs.orglett.6b00890
Penta‐2,4‐dien‐1‐ones by Formal [3+2] Cycloaddition–Rearrangement of Electron‐Deficient Diethyl 2‐(Dicyanomethylene)malonate with Alkynes

European Journal of Organic Chemistry, 2016, 716. 10.1002/ejoc.201501473
Flexible analogues of WAY-267,464: Synthesis and pharmacology at the human oxytocin and vasopressin 1a receptors

European Journal of Organic Chemistry, 2016, 108, 730. 10.1016/j.ejmech.2015.11.050
A systematic exploration of the effects of flexibility and basicity on sigma (σ) receptor binding in a series of substituted diamines
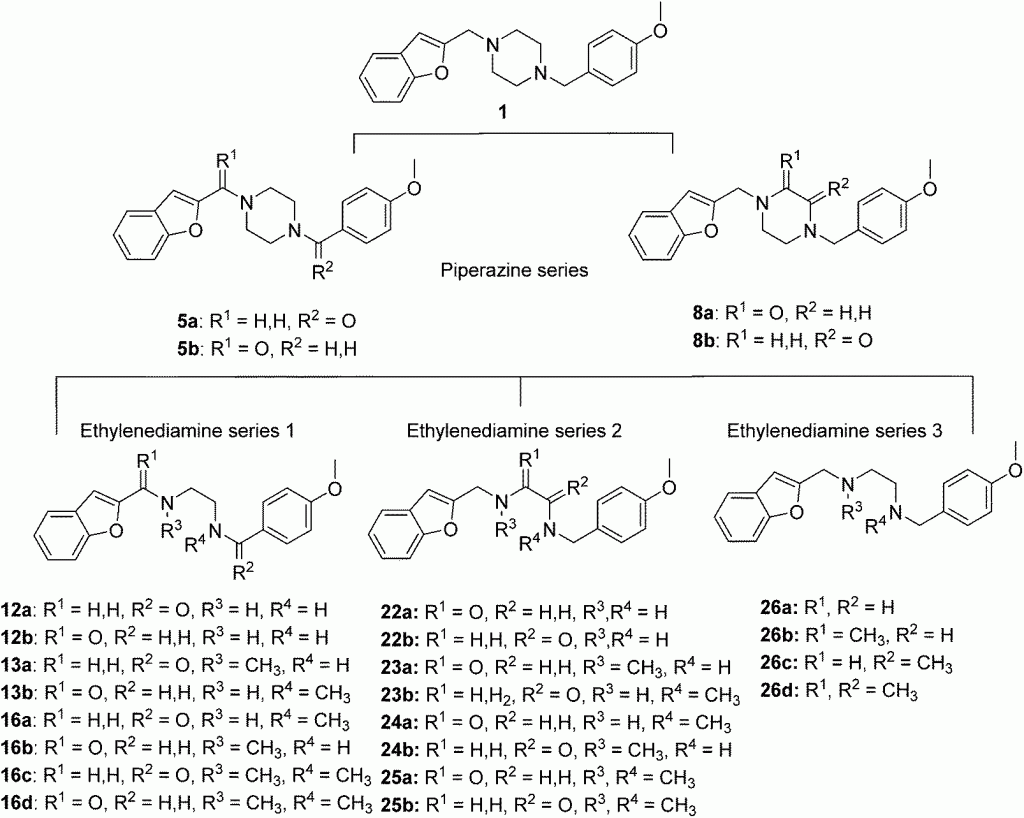
Organic & Biomolecular Chemistry, 2016, 14, 9388. 10.1039/C6OB00615A
Ester‐Substituted Electron‐Poor Alkenes for Cycloaddition–Retroelectrocyclization (CA–RE) and Related Reactions

European Journal of Organic Chemistry, 2015, 7264. 10.1002/ejoc.201501085
WAY 267,464, a non-peptide oxytocin receptor agonist, impairs social recognition memory in rats through a vasopressin 1A receptor antagonist action
Psychopharmacology, 2015, 232, 2659. 10.1007/s00213-015-3902-5
Structure–activity relationship studies of SEN12333 analogues: Determination of the optimal requirements for binding affinities at α7 nAChRs through incorporation of known structural motifs

European Journal of Organic Chemistry, 2015, 95, 277. 10.1016/j.ejmech.2015.03.025
Two-photon absorption and spectroscopy of the lowest two-photon transition in small donor-acceptor–substituted organic molecules
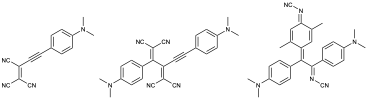
Physical Review A, 2015, 91, 043818. 10.1103/PhysRevA.91.043818
Investigations of amide bond variation and biaryl modification in analogues of α7 nAChR agonist SEN12333

European Journal of Medicinal Chemistry, 2014, 84, 200. 10.1016/j.ejmech.2014.07.029
Pyrazolo[1,4]diazepines as non-peptidic probes of the oxytocin and vasopressin receptors

Tetrahedron Letters, 2014, 55, 4568. 10.1016/j.tetlet.2014.06.022
Body temperature and cardiac changes induced by peripherally administered oxytocin, vasopressin and the non‐peptide oxytocin receptor agonist WAY 267,464: a biotelemetry study in rats
British Journal of Pharmacology, 2014, 171, 2868. 10.1111/bph.12613
RANEY® cobalt – an underutilised reagent for the selective cleavage of C–X and N–O bonds

Organic & Bioorganic Chemistry, 2014, 12, 7433. 10.1039/C4OB00917G
Synthesis of Biologically Active Seven-Membered-Ring Heterocycles
Synthesis, 2013, 45, 3211. 10.1055/s-0033-1338549
Intramolecular Cycloaddition Reactions of cis-1,2-Dihydrocatechol Derivatives Incorporating C3-Tethered Diazoketones, Nitrile Oxides, and Azides: Stereocontrolled Routes to Enantiomerically Pure Spiro[5.5]undecanes and Related Systems
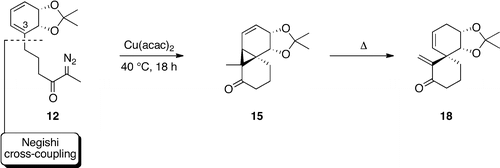
Journal of Organic Chemistry, 2013, 78, 7100. 10.1021/jo400952u
A Raney-Cobalt-Mediated Tandem Reductive Cyclization Route to the 1,5-Methanoazocino[4,3-b]indole Framework of the Uleine and StrychnosAlkaloids
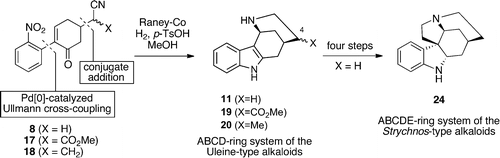
Journal of Organic Chemistry, 2012, 77, 10773. 10.1021/jo302132d
Application of a Raney-Cobalt-Mediated Tandem Reductive Cyclization Protocol to Total Syntheses of the Aspidosperma Alkaloids (±)-Limaspermidine and (±)-1-Acetylaspidoalbidine
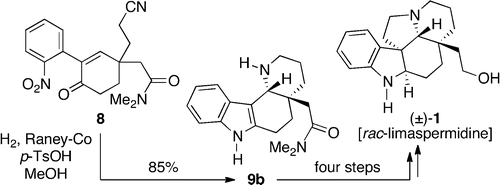
Organic Letters, 2012, 14, 5621. 10.1021/ol3026846
Chemoenzymatic Methods for the Enantioselective Assembly of Bioactive Natural Products
Chemistry in Australia, 2008, 75, 3.
The Chemoenzymatic Total Synthesis of Phellodonic Acid, a Biologically Active and Highly Functionalized Hirsutane Derivative Isolated from the Tasmanian Fungus Phellodon melaleucus
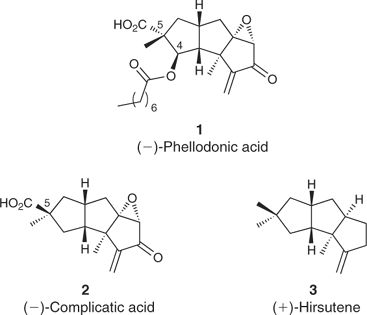
Australian Journal of Chemistry, 2008, 61, 94. 10.1071/CH07403
